Currently, cryoprotective agents (CPAs) such as glycerol, dimethyl sulfoxide (DMSO) and hydroxyethyl starch (HES) are primarily used in cryopreservation solutions. However, these CPAs do not protect against cell damage that ice crystals can cause when samples are inappropriately stored or undergo unwanted transient warming during handling, storage, and shipment. Additionally, these CPAs can be toxic and damage the cells that they are meant to preserve. Both events result in lower cell recovery, decreased cell viability, and impaired cell function.
Most of the cellular damage during cryopreservation occurs as a result of the uncontrolled growth of ice during storage at sub-zero temperatures or during subsequent warming and thawing. This process is known as ice recrystallization and ultimately results in decreased post-thaw viability and functional capacity of the cells and tissues that have been cryopreserved. Conventional CPAs do not protect against the process of ice recrystallization.
PanTHERA has developed a disruptive platform technology that improves the cryopreservation of biological material by controlling ice crystal size and growth throughout the cryopreservation process of cells and tissues. PanTHERA’s KryoAegisTM utilizes patented small molecules (< 300 AMUs) that function as ice recrystallization inhibitors (IRI). When added to traditional cryoprotective agents (CPAs) they can help create a more hospitable cellular environment. This can increase cellular recovery and quality, protect against damage from unintended warming, reduce the toxic effects of traditional CPAs and allow for higher sub-zero storage temperatures, thereby reducing the need for liquid nitrogen storage and shipping containers.
The application fields of PanTHERAs KryoAegisTM are extensive. These include:
-
-
-
- Research and development
- Gene and cell therapy products
- Organ, tissue, and blood banking
- Biomanufacturing industry (such as viral vectors, oncolytic virus, recombinant proteins, vaccines)
- Assisted reproductive technologies
- Animal and aquatic species breeding and preservation
-
-
Increasing Cellular Recovery and Quality
Human induced pluripotent stem cells (iPSCs) and iPSC-derived neurons (iPSC-Ns) represent a differentiated modality toward developing novel cell-based therapies for regenerative medicine. The cryopreservation of iPSC-Ns can be improved by adding KryoAegisTM in commercially available cryopreservation media.
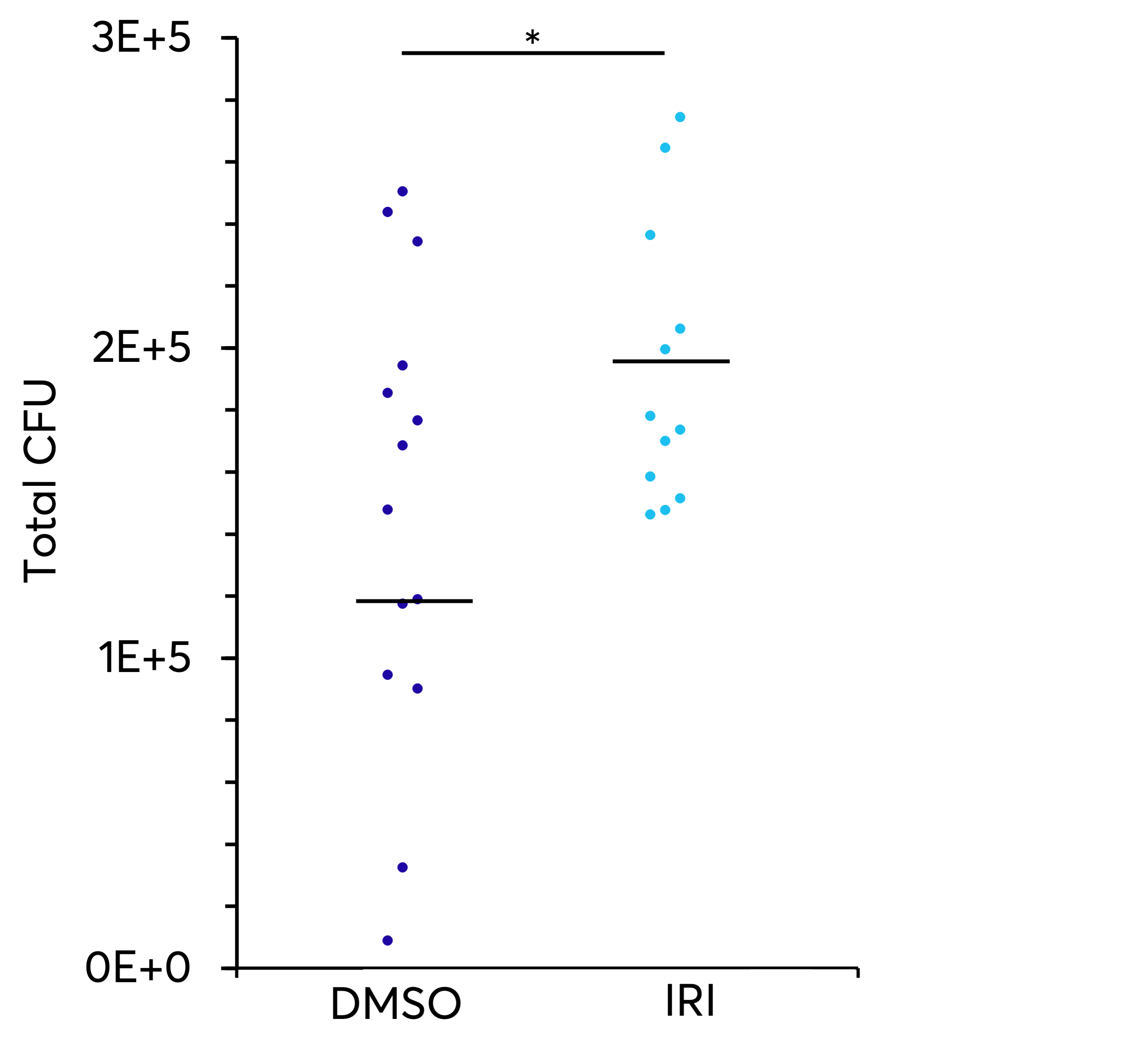
HSCs obtained from umbilical cord blood are typically cryopreserved using a slow cooling rate (1 °C/min) with a cryopreservation media containing 10% DMSO. The success of the transplant of these cells is directly related to the quantity and quality of the progenitor cells transplanted. A common problem for patients and healthcare systems is the slow engraftment or graft failure of these cells. The addition of KryoAegisTM to the cryopreservation media has demonstrated increased functionality of HSCs resulting in improved engraftment in a mouse model.
Protecting Against Unintentional Warming
Cryopreserved products will undergo periods during storage where the product temperature will transiently increase due to the logistics of retrieving products from bulk freezers, moving products between locations, preparing products for shipment, and compressor cycling or failure of equipment. This transient warming (repetitive cycling of temperature) of cryopreserved cells and tissues occurs during normal biobank operations and its impact on the storage stability and post-thaw quality of cryopreserved materials is a common problem seen in the industry. KryoAegisTM has the ability to protect cells from these transient warming events protecting cell quality.
The effect of transient warming was demonstrated on HUVEC cells that were stained with a nucleic acid dye (SYTO13) and cooled at 90 °C/min to –40 °C and warmed at 25 °C and –10 °C. As seen, the size of intracellular ice crystals imaged under fluorescence illumination, the size of the crystals as is significantly reduced in the presence of KryoAegisTM.
Moreover, the protection conferred by KryoAegisTM was demonstrated to also affect the post-thaw functionality of these cells. A tube formation assay mimics angiogenesis in vitro and is used to quantitate the ability of endothelial cells to form capillary-like tubular structures when cultured on reconstituted basement membrane. HUVEC were cryopreserved and stored at –80 °C and then repeatedly (x5) transiently warmed to –20 °C before assessment.
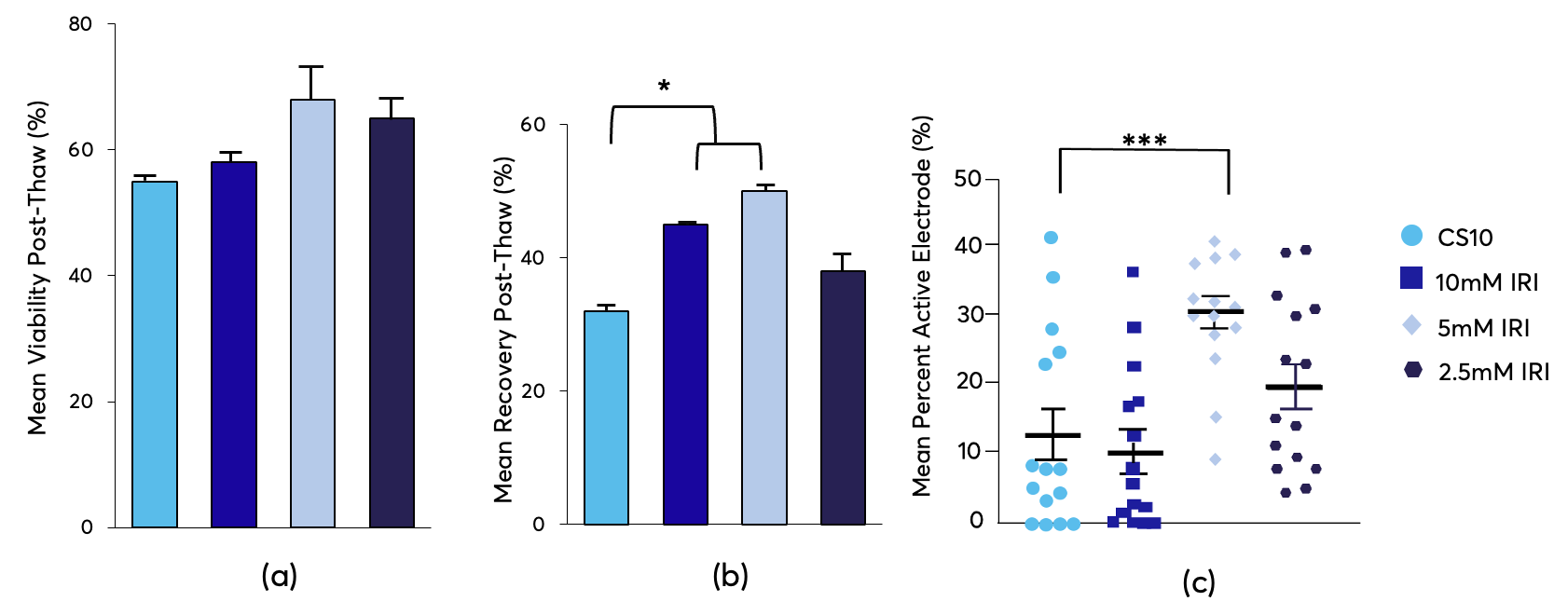
The addition of IRIs also protected red blood cells (RBC) from damaging warming events during cryopreservation in low concentrations of glycerol. Since IRIs controls ice growth during these warming events, which minimizes the ice crystals’ size, this results in increased viability after samples are thawed.
Reducing the Toxic Effects of DMSO
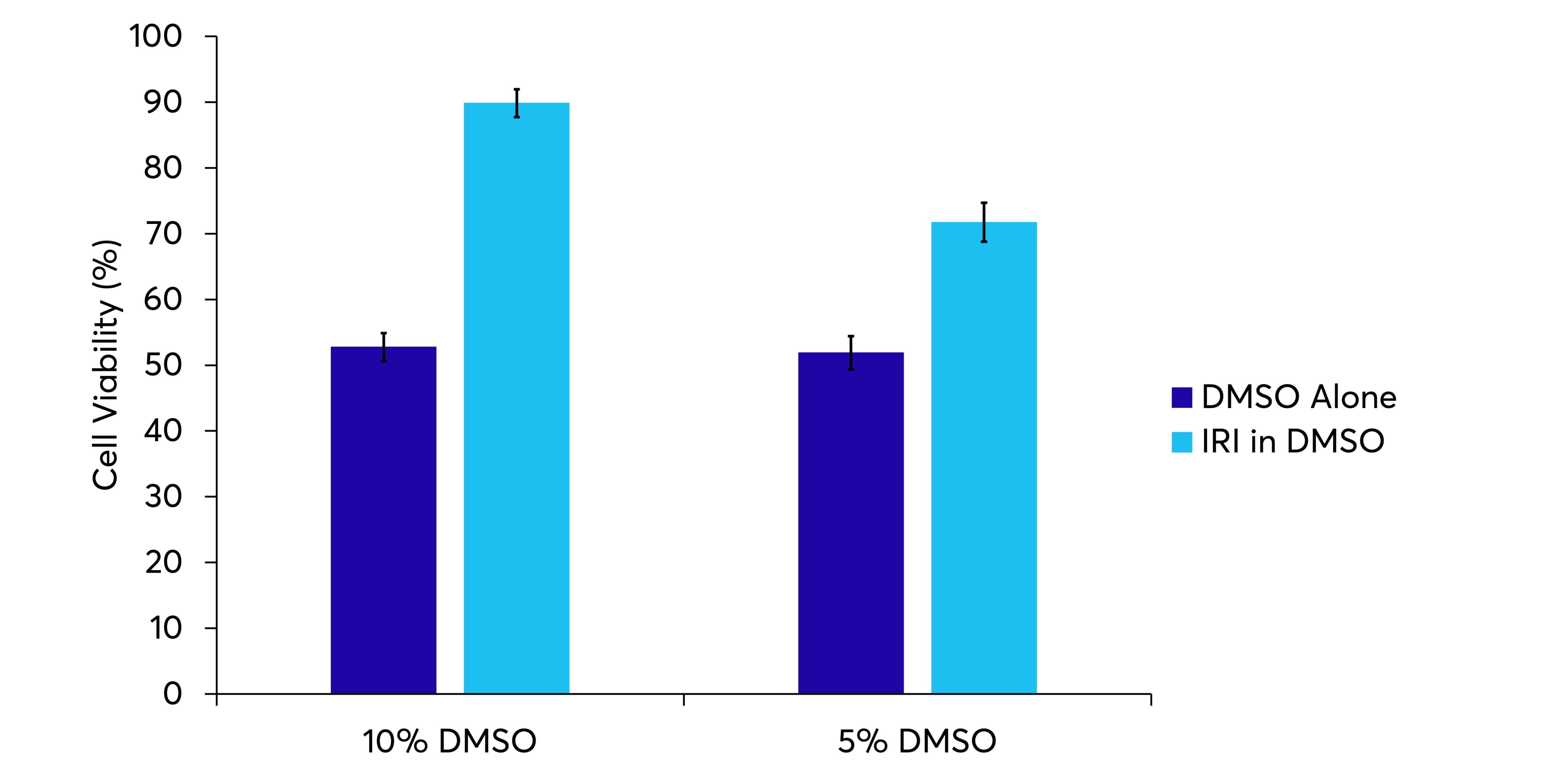
DMSO has cytotoxicity that can impact the quality, functionality, and survival of cells that have been cryopreserved. It has been reported that various concentrations of DMSO that are conventionally used (up to 10%) can reduce the response of cells in culture and limit survival rates of certain cell populations. By adding KryoAegisTM to cryopreservation media, a reduction in DMSO concentration can be achieved.
We’ve shown that using decreased DMSO concentrations in combination with KryoAegisTM allows for increased viability of BEAS-2B cells post-thaw when compared to decreasing concentrations of DMSO alone.
Other cell types, such as hepatocytes, also show an increase in cell viability when cells are frozen in reduced concentrations of DMSO in the presence of KryoAegisTM.
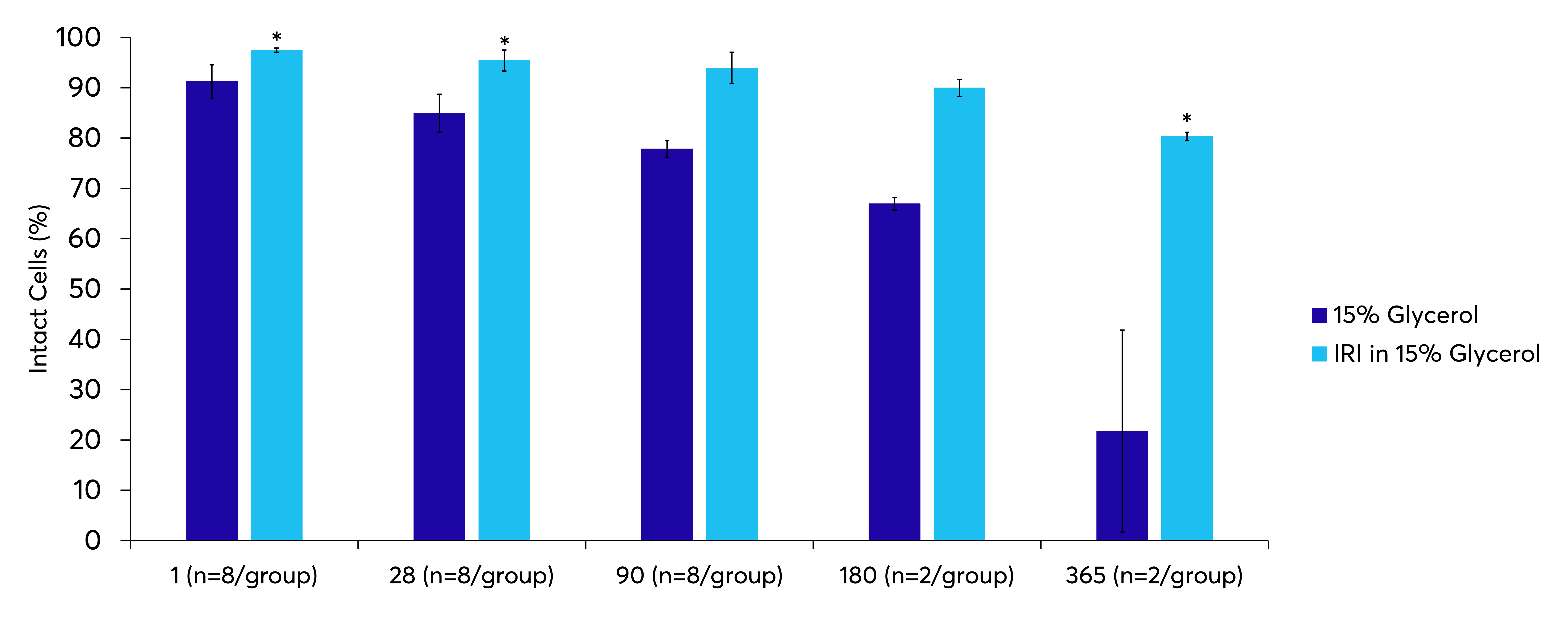
Stability at High Sub-Zero Temperatures
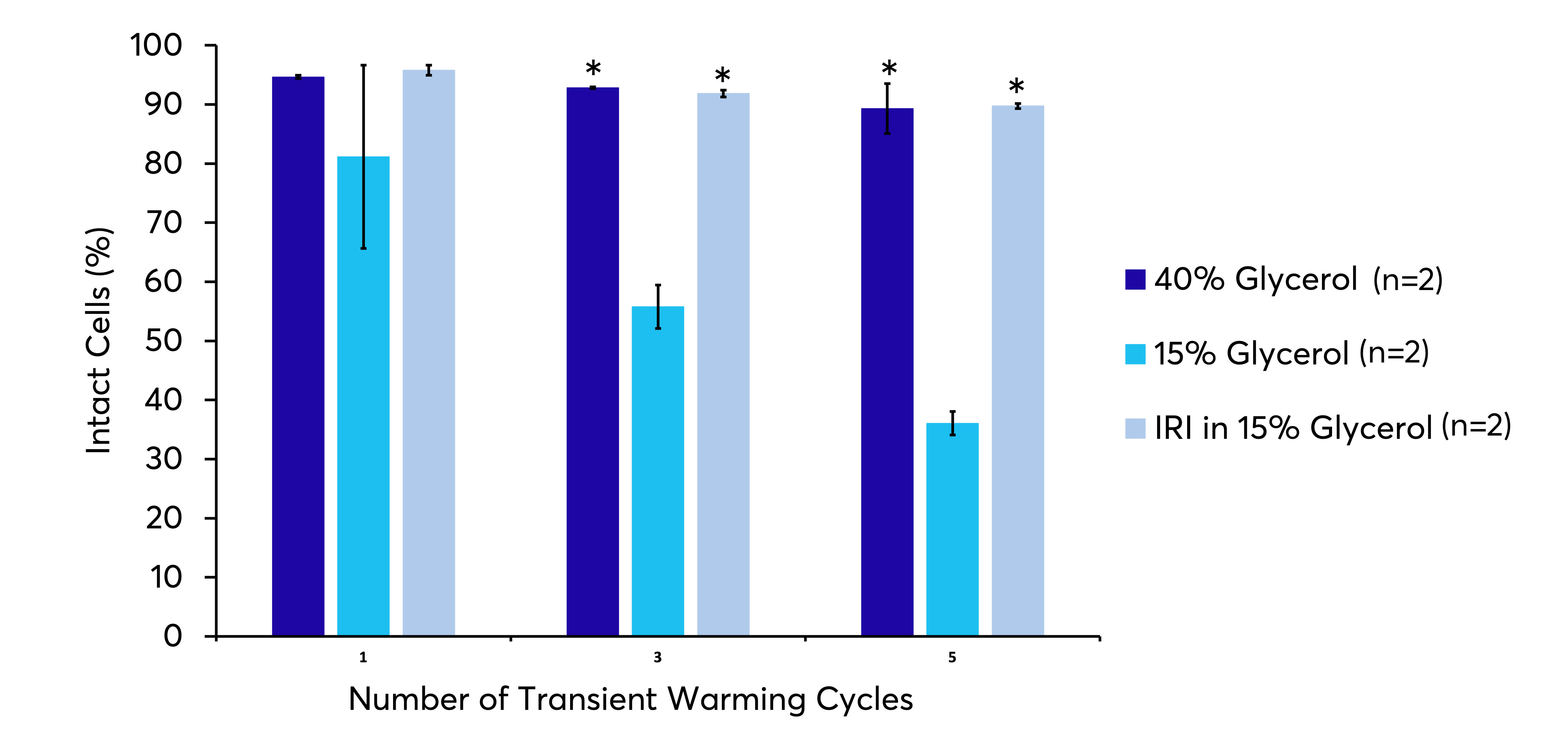
Conventional cryopreservation media has led to the storage of cellular products in the vapour or liquid phase of liquid nitrogen. A successful cryopreservation protocol will preserve the biological function of cells during the freezing and thawing processes. As temperature decreases, the complex mixture of solutes and solvents within the system will undergo a series of phase transitions (ie. freezing), ultimately resulting in the system reaching a fully solid state. The addition of IRI technology can allow for altered high sub-zero storage conditions extending the shelf life of products stored in more conventional freezers without the use of liquid nitrogen. RBCs cryopreserved with 15% glycerol must be stored in liquid nitrogen as they are highly susceptible to the damaging effects of transient warming. However, with the addition of an IRI, these cells can be stored in a conventional –80 °C freezer with greatly improved cellular recovery post- thaw when compared to glycerol alone.